K. Kinoshita1, S. Satake1,2,3, K. Murotani4, M. Takemura3,5, Y. Matsui3, H. Arai6
1. Department of Frailty Research, Center for Gerontology and Social Science, Research Institute, National Center for Geriatrics and Gerontology, Obu, Aichi, Japan; 2. Department of Geriatric Medicine, Hospital, National Center for Geriatrics and Gerontology, Obu, Aichi, Japan; 3. Center for Frailty and Locomotive Syndrome, Hospital, National Center for Geriatrics and Gerontology, Obu, Aichi, Japan; 4. Biostatistics Center, Kurume University, Kurume, Fukuoka, Japan; 5. Research & Development Promotion Office, Research Core Facilities, Research Institute, National Center for Geriatrics and Gerontology, Obu, Aichi, Japan; 6. National Center for Geriatrics and Gerontology, Obu, Aichi, Japan
Corresponding Author: Kaori Kinoshita, Department of Frailty Research, Center for Gerontology and Social Science, Research Institute, National Center for Geriatrics and Gerontology, 7-430, Morioka-cho, Obu, Aichi 474-8511, Japan, Phone: +81-0562-46-2311, Fax: +81-0562-46-2373, Email: kino4ta@ncgg.go.jp
J Frailty Aging 2022;11(4)393-397
Published online August 10, 2022, http://dx.doi.org/10.14283/jfa.2022.49
Abstract
The Frailty screening should be widely performed; however, simple and inexpensive biomarkers are missing. Biomarkers that can be routinely assessed in many patients are desirable. Recently, the hemoglobin-to-red cell distribution width ratio (Hb/RDW, HRR) has been suggested as a new prognostic marker and has been reported to be associated with inflammation, one of the factors contributing to frailty. Therefore, we aimed to address the role of HRR in frailty among 557 older outpatients (aged 65–96 years). Frailty was assessed using the Japanese version of the Cardiovascular Health Study criteria, and HRR was calculated from clinical records. Participants were classified into five groups based on a sex-stratified quintile of HRR (Q1–Q5). Of the participants, 20.3% were frail. Using multiple logistic regression models with the Q5 group as a reference, after adjusting for sex, age, body mass index, polypharmacy, pre-orthopedic surgery, and the use of iron medications, the multivariable-adjusted odds ratios (95% confidence intervals) of the Q4 to Q1 groups were 0.92 (0.58–1.47), 1.04 (0.67–1.61), 1.29 (0.84–1.96), and 1.85 (1.22–2.82), respectively, indicating that a lower HRR was significantly associated with frailty. The robustness of these results was also shown in the multiple imputation analysis. The results suggest that HRR measurement may be one of the indicators to identify frail older adults in routine practice.
Key words: Hemoglobin, red cell distribution width, frailty, biomarker, screening.
Introduction
Frailty screening should be performed for all older adults in daily practice. To achieve this goal, a biomarker obtained from a routine blood examination may be beneficial for the screening that identifies patients who should be assessed for frailty, including physical assessment. This is because blood examinations are conducted routinely and can be performed without requiring large space, expensive instruments, or additional cost.
A complete blood count (CBC) is a routine blood examination, and includes hemoglobin (Hb) and red cell distribution width (RDW). Both lower Hb levels and higher RDW have been reported to be associated with frailty (1-4), and the possible mechanism is systemic inflammation, which is one of the factors involved in the onset and progression of frailty (5, 6). Inflammation increases hepcidin level, which regulates iron metabolism and decreases blood iron levels, leading to lower Hb levels (7, 8). Further, inflammatory cytokines inhibit erythropoietin synthesis, shorten the erythrocytes lifespan, and increase the release of immature red blood cells, resulting in increased RDW (8-11).
Recently, Sun et al. suggested the peripheral Hb-to-RDW ratio (HRR) as a novel inflammatory marker (12). The first study on the ability of HRR to predict frailty was conducted by Qu et al. in 2021, in which they showed that HRR was more predictive for frailty than only Hb or RDW in hospitalized patients with coronary heart disease (10). However, to the best of our knowledge, no study has investigated the association between HRR and frailty other than the report by Qu et al. Therefore, it is necessary to investigate whether the association between HRR and frailty can be reproduced in different settings. We investigated the association between HRR and frailty in older outpatients.
Methods
Study design and participants
This cross-sectional study included participants of the Frailty Registry Study, which was conducted on outpatients of a frailty clinic in a general geriatric hospital in Aichi, Obu, Japan. In total, 595 participants without disabilities, which was defined with a Katz index score ≥5 (13), who visited the clinic between June 2017 and May 2020, were initially included. The eligibility criteria for the present study were as follows: (i) age ≥65 years and (ii) no history of erythropoietin administration. Finally, data from 557 participants were analyzed.
This study was approved by the Ethics Committee of Human Research of the National Center for Geriatrics and Gerontology, Japan (No. 881–11), and was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all study participants.
Frailty assessment
Physical frailty was assessed using the Japanese version of the Cardiovascular Health Study criteria (J-CHS), which has five components (shrinking, weakness, slowness, exhaustion, and low activity) and was based on the original criteria of the phenotype model proposed by Fried (14-16). The criteria were optimized for older Japanese adults and validated elsewhere (15-17). Frailty was defined as having three or more components, and pre-frailty was defined as having one or two components.
HRR calculation
Hb and RDW were obtained from the CBC, and the HRR was calculated by dividing Hb by RDW. Blood samples were collected at the first visit to a frailty clinic.
Covariates
Body mass index (BMI; kg/m2) was calculated using anthropometric data. Polypharmacy was defined as ≥5 medications based on clinical records. Participants with pre-orthopedic surgery and iron medication were identified from clinical records.
Statistical analysis
Continuous variables are presented as mean and standard deviation (SD), while categorical variables are presented as numbers and percentages (%). Participants were classified into five groups according to the sex-stratified quintile of HRR (Q1–Q5). The characteristics of the participants in these groups were compared using a one-way analysis of variance and the Jonckheere-Terpstra test for continuous variables, and the χ2 test for categorical variables.
Both complete-case and imputation-case analyses were analyzed. In the imputation-case analysis, the multiple imputation method was used to handle missing data with 20 imputed datasets using Rubin’s formula (18). In both complete-case and imputation-case analyses, with the Q5 group as reference, the associations between HRR and frailty, and between HRR and pre-frailty and frailty (i.e., J-CHS≥1) were analyzed using multiple logistic regression models. Covariates of model 1 were sex, age, BMI, and polypharmacy and those of model 2 were model 1 plus pre-orthopedic surgery, and the use of iron medications. Additionally, as a supplemental analysis, the associations between HRR (as a continuous variable) and five components of frailty were analyzed using multiple logistic regression models with the same covariates.
All statistical analyses were performed using the IBM SPSS Statistics ver. 28.0 (IBM Japan, Tokyo, Japan), and statistical significance was indicated by two-sided P-values<0.05.
Results
The missing values were as follows: gate speed (included in the five criteria of J-CHS), n=1; blood examination (including Hb and RDW), n=2. None of the covariate data was missing.
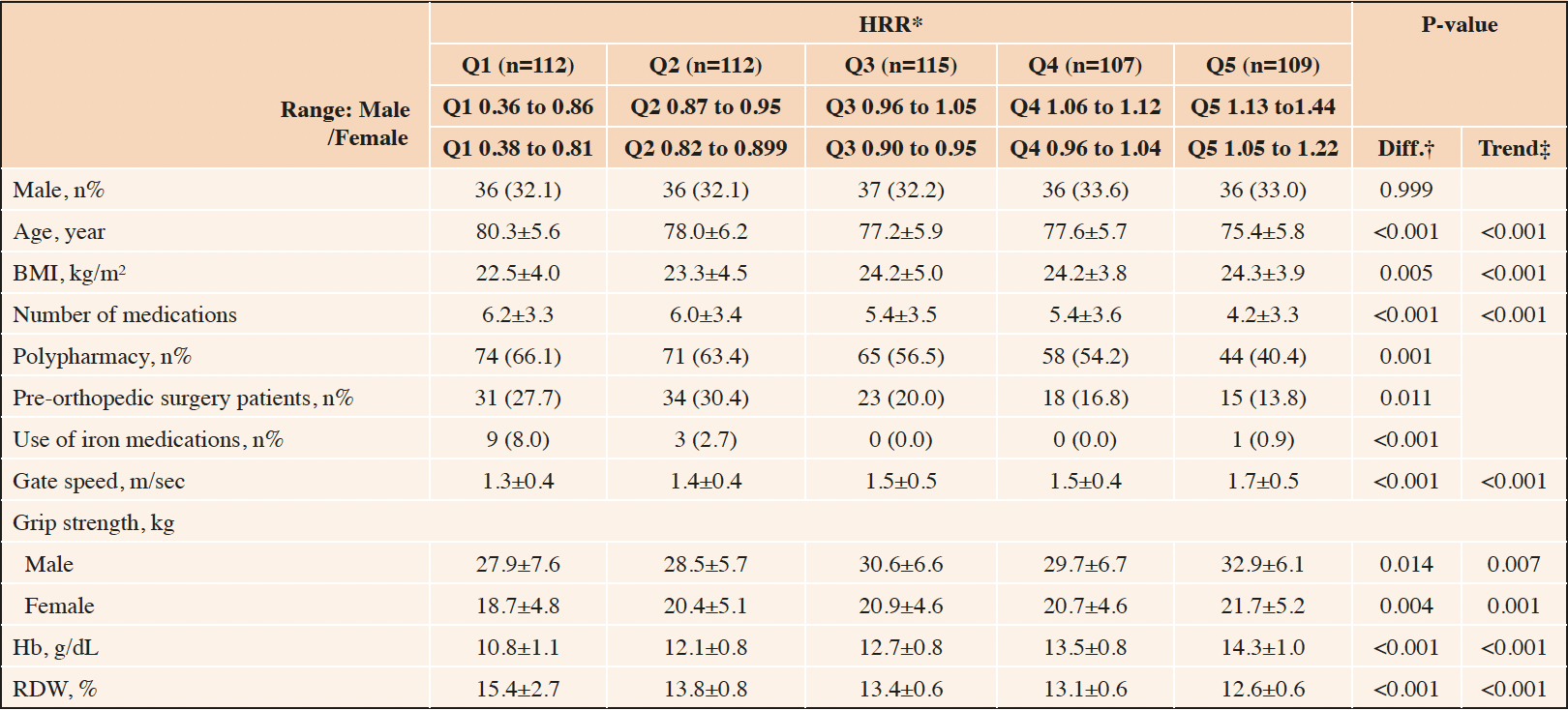
The mean ± SD (range) of age was 77.7 ± 6.0 (65–96) years. Among the 556 participants who had complete J-CHS data, 113 (20.3%) were frail and 328 (59.0%) were pre-frail. Participants were significantly younger, had a higher BMI, fewer medications, higher gate speed, and higher grip strength from the Q1 group to the Q5 group (Table 1).
*HRR = Hb/RDW; † P-values were obtained using one-way ANOVA for continuous variables and the χ2 test for categorical variables; ‡ P-values were obtained using the Jonckheere-Terpstra test; HRR, hemoglobin-to-red cell distribution width ratio; Q, quintile; Diff, difference; BMI, body mass index; Hb, hemoglobin; RDW, red cell distribution width; ANOVA, analysis of variance.
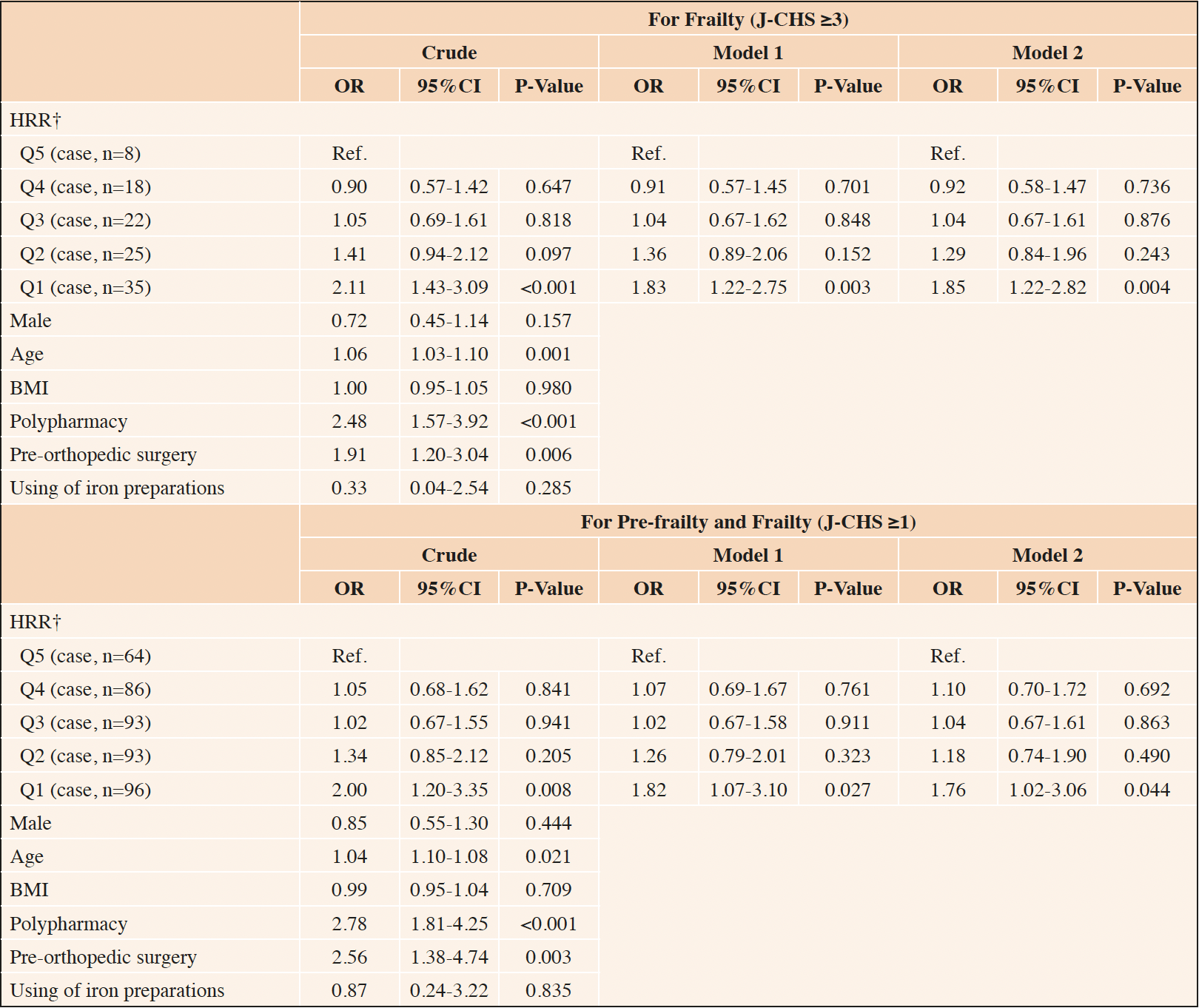
The multivariable-adjusted associations of HRR with pre-frailty and frailty are shown in Table 2. The OR for frailty was significantly higher in the Q1 group than in the Q5 group. This significant association was also observed for pre-frailty and frailty (i.e., J-CHS ≥1). The results of the multiple imputations showed the robustness of these complete case analyses.
*ORs and 95% CIs were estimated using the multiple logistic regression analysis. Model 1: adjusted for sex, BMI, and polypharmacy. Model 2: adjusted for pre-orthopedic surgery, and using of iron preparations in addition to the variables in model 1; †HRR = Hb/RDW; J-CHS, the Japanese version of the Cardiovascular Health Study criteria; OR, odds ratio; CI, confidence interval; HRR, hemoglobin–to–red cell distribution width ratio; Q, quintile; BMI, body mass index; Hb, hemoglobin; RDW, red cell distribution width.
In the supplemental analysis, the odds ratios (ORs) and 95% confidence intervals (CIs) of HRR for each of the five components of frailty were as follows: in model 1, shrinking 0.05 (0.01–0.21), P<0.001; exhaustion 0.83 (0.26–2.67), P=0.753; low activity 0.06 (0.02–0.21), P<0.001; slowness 0.09 (0.02–0.47), P=0.005; and weakness 0.28 (0.08–0.98), P=0.046. In model 2, shrinking 0.06 (0.01–0.24), P<0.001; exhaustion 0.51 (0.15–1.78), P=0.294; low activity 0.10 (0.03–0.36), P<0.001; slowness 0.06 (0.01–0.42), P=0.004; and weakness 0.06 (0.01–0.42), P=0.004. The robustness of these results was also shown in the multiple imputation analysis.
Discussion
This study clarified that a lower HRR was significantly associated with pre-frailty and frailty in older outpatients. This significance remained even after adjusting for polypharmacy and pre-orthopedic surgery, both of which had a significantly positive association with frailty in the univariate analysis. Polypharmacy and orthopedic diseases are common challenges in older patients, but these results suggest that HRR may be used to screen frailty independent of these problems. HRR can be calculated from Hb and RDW included in the CBC, which is a routine blood test, and is also suggested as a novel prognostic marker because it could theoretically reflect health conditions, including the nutritional status, inflammatory condition, and immune function (12). Our findings indicate that using HRR for frailty screening may be helpful for routine assessments.
This study had 20.3% frail older adults. It has been reported that the prevalence of frailty in Japanese community dwellers aged 75–79, 80–84, and ≥85 years is 10%, 20.4%, and 35.1%, respectively (19). The prevalence of frailty was slightly higher in this study than in recent studies, possibly because the present study was conducted on outpatients at a frailty clinic.
In the association between HRR and components of the frailty phenotype, HRR was significantly associated with four of five components, i.e., shrinking, low activity, slowness, and weakness, but was not associated with exhaustion. Exhaustion is also a main symptom of depression and has been suggested to be positively associated with frailty (20, 21). Furthermore, sleep disorders and poor sleep quality generally manifest as exhaustion, and an association between sleep problems and frailty has been reported (22, 23). Thus, HRR may not always reflect exhaustion due to other causes, such as depression and sleep disorders. However, Qu et al. suggested that HRR is the most optimal screening marker compared to only Hb or RDW, based on receiver operating characteristic analysis (10). Although lower Hb and anemia may decrease tissue oxygenation, reduce muscle synthesis and strength, and cause frailty (24, 25), previous studies have reported sex differences in the association between Hb and frailty (26, 27). Of note, RDW is considered an indicator of cellular senescence, i.e., telomere shortening (28) and a marker of inflammation (11). Thus, HRR, which is a combined index of Hb and RDW, may reflect frailty more strongly than each marker alone. It is reasonable to routinely check HRR for screening individuals for frailty at outpatient clinics.
The present study has some limitations. First, this was a single-center study, and the participants were limited to outpatients of a frailty clinic in Japan. Future studies in other settings are needed. Second, we investigated the association between HRR and frailty based on a cross-sectional study to consider whether HRR can be used to screen people who should be assessed for frailty. It is important to investigate the predictive ability of HRR for adverse outcomes in older people with a longitudinal study. Third, 13 of the participants in this study had taken iron medications. Given that iron medications increase Hb and decrease RDW (29), the HRR of those taking iron pills may be higher than the actual HRR level. Thus, the association between HRR and frailty in those participants may have been overestimated.
In conclusion, a lower HRR was significantly associated with pre-frailty and frailty based on the phenotype model among older Japanese outpatients. This suggests that using HRR to screen people who require a detailed evaluation for frailty, that is, using HRR as the first step for routine frailty screening, would provide an opportunity for frailty assessment in many older people.
Acknowledgments: The authors would like to thank all study participants and staff of the frailty clinic at our institute for their cooperation and contribution to this study. We also thank Editage (www.editage.com) for the English language editing.
Conflicts of interest: The authors declare no conflicts of interest in the present study.
Authors’ contributions: Kaori Kinoshita, Conceptualization, Formal analysis, Methodology, Writing-original draft, and Writing-review & editing; Shosuke Satake, Conceptualization, Funding acquisition, Investigation, Methodology, and Writing-review & editing; Kenta Murotani, Conceptualization, Formal analysis, Methodology, and Writing-review & editing; Marie Takemura, Conceptualization, Methodology, and Writing-review & editing; Yasumoto Matsui, Conceptualization, Data curation, Funding acquisition, Investigation, Methodology, Project administration, and Writing-review & editing; Hidenori Arai, Conceptualization, Investigation, Methodology, Project administration, Supervision, and Writing-review & editing; All authors critically revised the manuscript for intellectual content and approved the final version.
Sources of support: This study was supported in part by Research Funding for Longevity Sciences from the National Center for Geriatrics and Gerontology, Japan (grant number 20–12), and the Japan Health Research Promotion Bureau under Research (grant number 2021-B01). No grant provider played any role in the design, analysis, or writing of this manuscript.
Ethical standards: This study was approved by the Ethics Committee of Human Research of the National Center for Geriatrics and Gerontology, Japan (No. 881–11), and was conducted in accordance with the Declaration of Helsinki. .
Open Access: This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, duplication, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
References
1. Mailliez A, Guilbaud A, Puisieux F, Dauchet L, Boulanger É. Circulating biomarkers characterizing physical frailty: CRP, hemoglobin, albumin, 25OHD and free testosterone as best biomarkers. Results of a meta-analysis. Exp Gerontol 2020;139:111014. https://doi.org/10.1016/j.exger.2020.111014.
2. Picca A, Coelho-Junior HJ, Calvani R, Marzetti E, Vetrano DL. Biomarkers shared by frailty and sarcopenia in older adults: a systematic review and meta-analysis. Ageing Research Reviews 2022;73:101530. https://doi.org/10.1016/j.arr.2021.101530.
3. Li CM, Chao CT, Chen SI, Han DS, Huang KC. Elevated red cell distribution width is independently associated with a higher frailty risk among 2,932 community-dwelling older adults. Front Med (Lausanne) 2020;7:470. https://doi.org/10.3389/fmed.2020.00470.
4. Kim KM, Lui LY, Browner WS, et al. Association between variation in red cell size and multiple aging-related outcomes. J Gerontol A Biol Sci Med Sci 2021;76:1288–1294. https://doi.org/10.1093/Gerona/glaa217.
5. Hoogendijk EO, Afilalo J, Ensrud KE, Kowal P, Onder G, Fried LP. Frailty: implications for clinical practice and public health. Lancet 2019;394:1365–1375. https://doi.org/10.1016/s0140-6736(19)31786-6.
6. Ferrucci L, Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol 2018;15:505–522. https://doi.org/10.1038/s41569-018-0064-2.
7. Nemeth E, Ganz T. Anemia of inflammation. Hematol Oncol Clin North Am 2014;28:671–681, vi. https://doi.org/10.1016/j.hoc.2014.04.005.
8. Straat M, van Bruggen R, de Korte D, Juffermans NP. Red blood cell clearance in inflammation. Transfus Med Hemother 2012;39:353–361. https://doi.org/10.1159/000342229.
9. Jelkmann W. Proinflammatory cytokines lowering erythropoietin production. J Interferon Cytokine Res 1998;18:555–559. https://doi.org/10.1089/jir.1998.18.555.
10. Qu J, Zhou T, Xue M, et al. Correlation analysis of hemoglobin-to-red blood cell distribution width ratio and frailty in elderly patients with coronary heart disease. Front Cardiovasc Med 2021;8:728800. https://doi.org/10.3389/fcvm.2021.728800.
11. Ferrucci L, Guralnik JM, Woodman RC, et al. Proinflammatory state and circulating erythropoietin in persons with and without anemia. Am J Med 2005;118:1288. https://doi.org/10.1016/j.amjmed.2005.06.039.
12. Sun P, Zhang F, Chen C, et al. The ratio of hemoglobin to red cell distribution width as a novel prognostic parameter in esophageal squamous cell carcinoma: a retrospective study from southern China. Oncotarget 2016;7:42650–42660. https://doi.org/10.18632/oncotarget.9516.
13. Katz S, Ford AB, Moskowitz RW, Jackson BA, Jaffe MW. Studies of illness in the aged. The index of adl: a standardized measure of biological and psychosocial function. Jama 1963;185:914–919. https://doi.org/10.1001/jama.1963.03060120024016.
14. Fried LP, Tangen CM, Walston J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 2001;56:M146–M156. https://doi.org/10.1093/gerona/56.3.m146.
15. Satake S, Shimada H, Yamada M, et al. Prevalence of frailty among community-dwellers and outpatients in Japan as defined by the Japanese version of the Cardiovascular Health Study criteria. Geriatr Gerontol Int 2017;17:2629–2634. https://doi.org/10.1111/ggi.13129.
16. Satake S, Arai H. The revised Japanese version of the Cardiovascular Health Study criteria (revised J-CHS criteria). Geriatr Gerontol Int 2020;20:992–993. https://doi.org/10.1111/ggi.14005.
17. Makizako H, Shimada H, Doi T, Tsutsumimoto K, Suzuki T. Impact of physical frailty on disability in community-dwelling older adults: a prospective cohort study. BMJ Open 2015;5:e008462. https://doi.org/10.1136/bmjopen-2015-008462.
18. Rubin DB, Schenker N. Multiple imputation in health-care databases: an overview and some applications. Stat Med 1991;10:585–598. https://doi.org/10.1002/sim.4780100410.
19. Kojima G, Iliffe S, Taniguchi Y, Shimada H, Rakugi H, Walters K. Prevalence of frailty in Japan: A systematic review and meta-analysis. J Epidemiol 2017;27:347–353. https://doi.org/10.1016/j.je.2016.09.008.
20. Pearson E, Siskind D, Hubbard RE, Gordon EH, Coulson EJ, Warren N. Frailty and severe mental illness: a systematic review and narrative synthesis. J Psychiatr Res 2022;147:166–175. https://doi.org/10.1016/j.jpsychires.2022.01.014.
21. Vasquez-Goñi GAJ, Papuico-Romero BM, Urrunaga-Pastor D, Runzer-Colmenares FM, Parodi JF. The depressed frail phenotype as a risk factor for mortality in older adults: a prospective cohort in Peru. Heliyon 2022;8:e08640. https://doi.org/10.1016/j.heliyon.2021.e08640.
22. Liu M, Hou T, Nkimbeng M, et al. Associations between symptoms of pain, insomnia and depression, and frailty in older adults: a cross-sectional analysis of a cohort study. Int J Nurs Stud 2021;117:103873. https://doi.org/10.1016/j.ijnurstu.2021.103873.
23. Shih AC, Chen LH, Tsai CC, Chen JY. Correlation between sleep quality and frailty status among middle-aged and older Taiwanese people: a community-based, cross-sectional study. Int J Environ Res Public Health 2020;17. https://doi.org/10.3390/ijerph17249457.
24. Artz AS. Anemia and the frail elderly. Semin Hematol 2008;45:261–266. https://doi.org/10.1053/j.seminhematol.2008.06.002.
25. Dodd SL, Powers SK, Brooks E, Crawford MP. Effects of reduced O2 delivery with anemia, hypoxia, or ischemia on peak VO2 and force in skeletal muscle. J Appl Physiol (1985) 1993;74:186–191. https://doi.org/10.1152/jappl.1993.74.1.186.
26. Li Q, Chen X, Han B. Effect modification by sex of the hemoglobin concentration on frailty risk in hospitalized older patients. Clin Interv Aging 2021;16:687–696. https://doi.org/10.2147/cia.S298672.
27. Denfeld QE, Habecker BA, Camacho SA, et al. Characterizing sex differences in physical frailty phenotypes in heart failure. Circ Heart Fail 2021;14:e008076. https://doi.org/10.1161/circheartfailure.120.008076.
28. Kozlitina J, Garcia CK. Red blood cell size is inversely associated with leukocyte telomere length in a large multi-ethnic population. PLoS One 2012;7:e51046. https://doi.org/10.1371/journal.pone.0051046.
29. Yokoyama K, Fukagawa M, Akiba T, et al. Randomised clinical trial of ferric citrate hydrate on anaemia management in haemodialysis patients with hyperphosphataemia: ASTRIO study. Sci Rep 2019;9:8877. https://doi.org/10.1038/s41598-019-45335-4.