C.S. Kow1, S.S. Hasan2,3
1. School of Postgraduate Studies, International Medical University, Kuala Lumpur, Malaysia; 2. School of Applied Sciences, University of Huddersfield, Huddersfield, United Kingdom; 3. School of Biomedical Sciences & Pharmacy, University of Newcastle, Callaghan, Australia
Corresponding Author: Chia Siang Kow, International Medical University, Kuala Lumpur, Malaysia, Email ID: chiasiang_93@hotmail.com
Dear Editor,
We read with great interest the consensus recommendations issued by Boreskie et al. (1) to prevent older adults from frailty progression and health declines during COVID-19 pandemic. The effort by authors to disseminate recommendations which had been summarized in the SAVE model is commendable since it serves to remind the clinicians not to neglect the frail older individuals even amid strained health care resources from the COVID-19 pandemic. In fact, we agree with authors that these strategies should be implemented to maintain and/or improve the functional status of frail individuals since frail individuals may be at risk for a worse prognosis shall they acquire COVID-19. In addition, due to declined health state, frail individuals may be more susceptible to the acquisition of COVID-19 (2). Our preliminary investigation had demonstrated that the frail individuals have been overrepresented among population with COVID-19, with the following are illustrative of our findings.
We perform a comprehensive literature search in PubMed, Scopus, Google Scholar, and preprint repositories (medRxiv and Research Square) from December 1, 2019 up to November 26, 2020, using the following keywords which included “COVID-19” or “SARS-CoV-2” or “severe acute respiratory syndrome coronavirus 2” and “frailty” or “frail” with no language restriction. Studies eligible for inclusion were those with any design, assessed frailty with any validated frailty assessment tools, and reported prevalence of frailty in patients with COVID-19. We excluded article types such as comments, narrative reviews, conference papers, and case reports without reporting original data.
After removing duplicates, pair of reviewers (CSK and SSH) independently reviewed the titles and abstracts to identify the articles meeting eligibility criteria. The full-text screening was subsequently used to identify a final list of studies that corresponded to the inclusion and exclusion criteria. If multiple studies were available from the same cohort of patients, the study with the largest sample was included in the review. Disagreement between the two reviewers was resolved through discussions. Two investigators (CSK and SSH) independently extracted relevant data from included studies: family name of the first author, publication year, study design, study setting (single centre, multicentre, or database review), age of participants, sample size, the prevalence of frailty, and frailty assessment scale. We employed a random-effects model to estimate the pooled prevalence of frailty and 95% confidence interval (CI). We examined the heterogeneity between studies using the I2 statistics with 50% and using the χ2 test with P <0.10, as the thresholds for statistically significant heterogeneity.
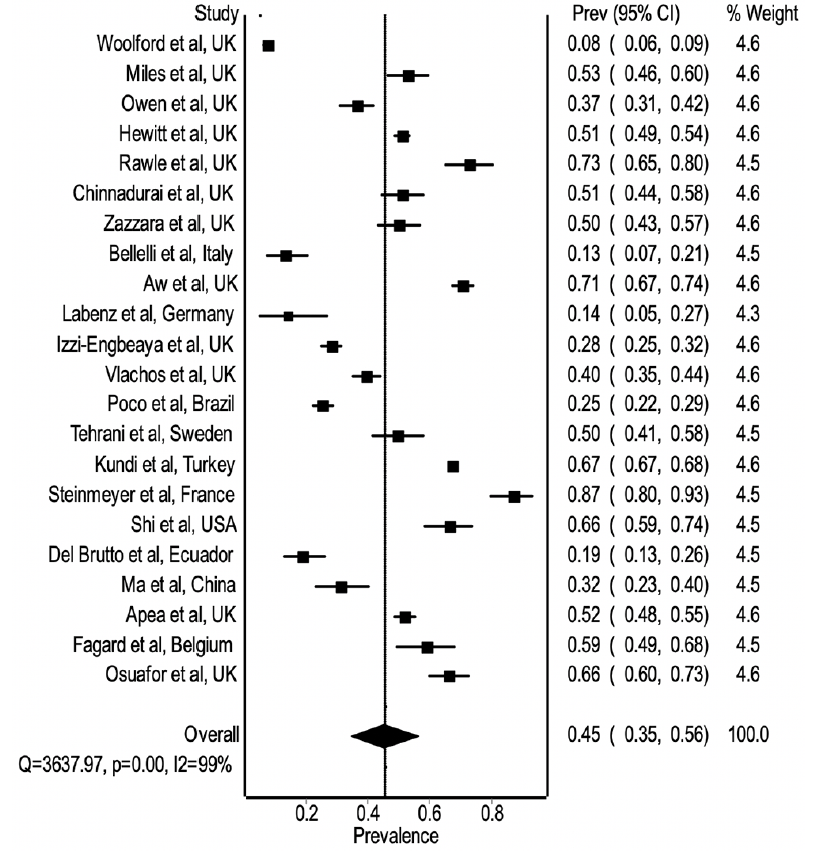
In total, 22 studies (list of reference as Supplementary Data) representing data from 25,246 patients with COVID-19 were included. Majority of the studies were from the European countries (the United Kingdom [n=12], Italy [n=1], Germany [n=1], Sweden [n=1], France [n=1], Belgium [n=1], Turkey [n=1]), while the remaining studies were from Brazil (n=1), the United States of America [n=1], Ecuador [n=1], and China [n=1]. The pooled prevalence of frailty across these 22 studies in a random-effect model of meta-analysis was 45% (Figure 1; 95% CI 35% to 56%; I2=99%; P <0.001). The pooled prevalence of frailty was much higher than the recent meta-analysis by O’Caoimh et al. (3) which reported an overall estimated worldwide frailty prevalence of 18% (95% CI 17% to 19%) among community-dwelling adults.
Such indirect comparison has indicated that frail individuals may be overrepresented among the COVID-19 patient population and given a rather strong hint that the presence of frailty may lead to a higher risk of acquisition of COVID-19. Therefore, the strategies put forth by Boreskie et al. (1)to overcome frailty and its associated complications become more important than before amid the COVID-19 pandemic since frailty could also predict a worse prognosis in patients with COVID-19. Furthermore, the frail individuals should be prioritized for prophylactic measures against COVID-19, either pharmacologically or non-pharmacologically. Administration of vitamin D proposed by authors as one of the measures to tackle frailty may in fact kill two birds with one stone: vitamin D could lead to improvement in the physical performance among older adults (4), and may be beneficial to prevent poor disease outcomes shall patients acquire COVID-19, since vitamin D deficiency (as suggested by serum 25 (OH)D concentration <20 ng/mL) had been associated with a severe course of COVID- 19 (5). Last but not least, the fact that almost 1 in 2 patients with COVID-19 was frail should alert the clinicians for standardized screening of frailty status in this patient population, at least among older individuals (≥60 years of age). Nevertheless, a limitation of our meta-analysis is related to the timing of the assessment of frailty in the included studies. Since the assessment of frailty was performed at baseline upon hospital admission, the prevalence of frailty could have been overestimated in these studies. Therefore, future studies could perform assessment of frailty in a longitudinal manner, without limited to assessment upon admission.
Conflict of interest: CSK and SSH have no conflicts of interest to declare.
References
1. Boreskie KF, Hay JL, Duhamel TA. Preventing Frailty Progression during the COVID-19 Pandemic. J Frailty Aging. 2020;9(3):130-131.
2. Xue QL. Frailty as an integrative marker of physiological vulnerability in the era of COVID-19. BMC Med. 2020;18(1):333.
3. O’Caoimh R, Sezgin D, O’Donovan MR, et al. Prevalence of frailty in 62 countries across the world: a systematic review and meta-analysis of population-level studies [published online ahead of print, 2020 Oct 17]. Age Ageing. 2020;afaa219.
4. Ju SY, Lee JY, Kim DH. Low 25-hydroxyvitamin D levels and the risk of frailty syndrome: a systematic review and dose-response meta-analysis. BMC Geriatr. 2018;18(1):206.
5. Jain A, Chaurasia R, Sengar NS, Singh M, Mahor S, Narain S. Analysis of vitamin D level among asymptomatic and critically ill COVID-19 patients and its correlation with inflammatory markers. Sci Rep. 2020;10(1):20191.